
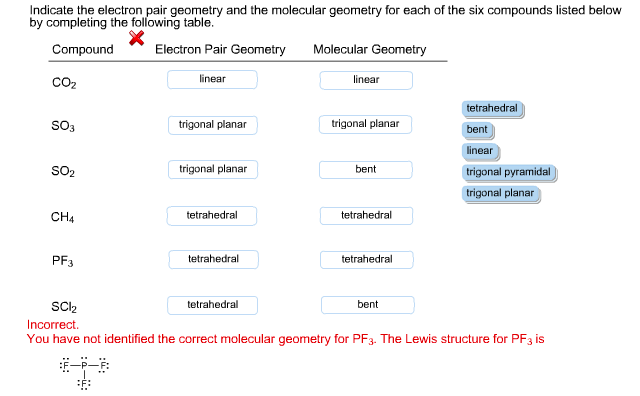
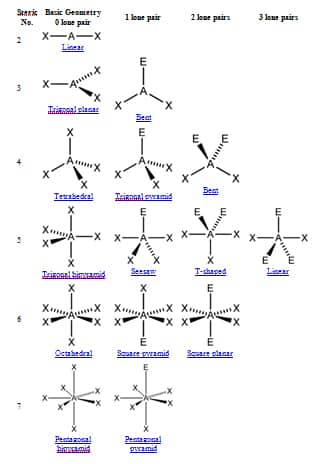
In this molecule, Phosphorus has one lone pair of electrons along with three bonding pairs of electrons. The bond angles are approximately 93.5° for the PH3 molecule. The central atom forms three sigma bonds with Hydrogen atoms and has a lone pair of electrons. PH3 Bond AnglesĪlthough Phosphine or PH3 molecule resemble NH3 molecule, there is a difference in their bond angles. Because it is a Drago molecule, there is no hybridization instead, pure p-orbitals participate in bond formation. As a result, there is no hybridization for the PH3 molecule. Apart from this, it also belongs to group 15. Here in PH3, the electronegativity value for Phosphorus is 2.19 ( less than 2.5), and it also has one lone pair of electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
